
Bioaccumulation and impact of copper pyrithione impact in juveniles of rainbow trout
 based on reviews by Anne-Sophie Voisin and 1 anonymous reviewer
based on reviews by Anne-Sophie Voisin and 1 anonymous reviewer
Characterization of the bioaccumulation and toxicity of copper pyrithione, an antifouling compound, on juveniles of rainbow trout
Abstract
Recommendation: posted 25 September 2023, validated 25 September 2023
Cosio, C. (2023) Bioaccumulation and impact of copper pyrithione impact in juveniles of rainbow trout. Peer Community in Ecotoxicology and Environmental Chemistry, 100104. 10.24072/pci.ecotoxenvchem.100104
Recommendation
Our ability to anticipate and estimate how pollution affects biota is intrumental in the field of ecotoxicology. Impact of chemical pollution by metals, drugs or pesticides was widely studied in different species using acute and chronic scenarios. Since the ban on tributyltin in antifouling paints, other copper (Cu)-based paints are on the market, including a new generation of booster biocides:metal pyrithiones such as Cu pyrithione (CuPT). Pyrithione acts as a Cu ionophore facilitating Cu transport across the membranes. Although some data show their occurrence in aquatic ecosystems and few studies on the toxicity of CuPT in fish are published, major gaps in knowledge remain about their toxicity and toxic pathway. Few studies were previously conducted in animals exposed to CuPT pointing to reprotoxicity, developmental malformation and mortality (Li et al. 2021, Mochida et al., 2011; Mohamat-Yusuff et al., 2018, Shin et al., 2022). However, its toxicokinetic and toxicodynamic remain to be characterized in details.
In this context, Bourdon et al. (2023) compared in juveniles of rainbow trout (Oncorhynchus mykiss), the effects of exposure to CuPT and ionic Cu2+ at equivalent Cu2+ molar concentrations. Presented data allow to compare the toxicity threshold, the accumulation of Cu and mechanisms of toxicity of both compounds. Acute and chronic exposures showed a higher bioaccumulation of Cu in the gills, and a higher toxicity of CuPT than that of ionic Cu2+, e.g. mortality , transcription levels of genes related to oxidative stress, detoxification and Cu transport. Intriguingly, the activities of enzymatic biomarkers used as proxy of antioxidant capacity were not significantly altered, although Cu is generally expected to trigger oxidative stress. In conlusion, this study brings new knowledge pointing that the presence of CuPT in the environment could induce toxic effects in non-target species. Moreover, it support the need to study in detail the toxicity of Cu-based paints to adapt regulations concerning their use and release in aquatic environments. Because of its low solubility in water, CuPT is expected to adsorb to suspended matter and food pellets. Future research should study this route of exposure.
References
Bourdon, C., Cachot, J., Gonzalez, P., Couture, P., 2023. Characterization of the bioaccumulation and toxicity of copper pyrithione, an antifouling compound, on juveniles of rainbow trout, bioRxiv ver. 3 peer-reviewed and recommended by Peer Community in Ecotoxicology and Environmental Chemistry. https://doi.org/10.1101/2023.01.31.526498
Li, X., S. Ru, H. Tian, S. Zhang, Z. Lin, M. Gao and J. Wang, 2021. Combined exposure to environmentally relevant copper and 2,2′-dithiobis-pyridine induces significant reproductive toxicity in male guppy (Poecilia reticulata). Science of the Total Environment 797, https://doi.org/10.1016/j.scitotenv.2021.149131
Mochida, K., Amano, H., Onduka, T., Kakuno, A., Fujii, K., 2011. Toxicity and metabolism of copper pyrithione and its degradation product, 2,2’-dipyridyldisulfide in a marine polychaete. Chemosphere 82, 390–397, https://doi.org/10.1016/j.chemosphere.2010.09.074
Mohamat-Yusuff, F., Sarah-Nabila, Ab.G., Zulkifli, S.Z., Azmai, M.N.A., Ibrahim, W.N.W., Yusof, S., Ismail, A., 2018. Acute toxicity test of copper pyrithione on Javanese medaka and the behavioural stress symptoms. Marine Pollution Bulletin 127, 150–153, https://doi.org/10.1016/j.marpolbul.2017.11.046
Shin, D., Y. Choi, Z. Y. Soon, M. Kim, D. J. Kim and J. H. Jung, 2022. Comparative toxicity study of waterborne two booster biocides (CuPT and ZnPT) on embryonic flounder (Paralichthys olivaceus). Ecotoxicology and Environmental Safety 233, https://doi.org/10.1016/j.ecoenv.2022.113337
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article. The authors declared that they comply with the PCI rule of having no financial conflicts of interest in relation to the content of the article.
This study was funded by a Discovery grant from the Natural Sciences and Engineering Research Council of Canada to P. Couture and by funds from the EPOC laboratory.
Reviewed by Anne-Sophie Voisin, 04 Sep 2023
I appreciate the changes made by the authors, who carefully replied to all the comments made for the first round of review. I am looking forward to see the article published.
Evaluation round #1
DOI or URL of the preprint: https://doi.org/10.1101/2023.01.31.526498
Version of the preprint: 2
Author's Reply, 28 Jul 2023
Dear Recommender,
I have attached a detailed reply to reviewer's file, along with the revised manuscript in track changes mode. The clean revised PDF is available on BioRxiv.
Decision by Claudia Cosio , posted 02 Jun 2023, validated 16 Jun 2023
, posted 02 Jun 2023, validated 16 Jun 2023
Dear authors,
I apologize for the delay: it was difficult to find reviewers. Based on the comments, I recommend major revision. Please adress all reviewer comments in detail.
Best regards,
Reviewed by anonymous reviewer 1, 03 May 2023
Characterization of the bioaccumulation and toxicity of copper pyrithione, an antifouling compound, on juveniles of rainbow trout.
General comments
This study presents a classical approach to evaluate bioaccumulation and toxic effects of copper pyrithione on rainbow trout juveniles Oncorhynchus mykiss. Original comparison was made with Cu2+ effects through CuSO4 exposure, in equivalent Cu2+ molar concentrations. Two laboratory experiments were conducted: a 24h acute exposure followed by a 16 days chronic exposure. Mortality, growth, copper accumulation, antioxidant enzymes activity and expression of genes involved in various functions were measured. Results revealed a higher toxicity of CuPT than that of ionic Cu from CuSO4, as observed through mortality and genes expression modulation. This study responds to the need of improve knowledge on toxicity of antifouling that are used as alternative to prohibited ones.
The experimental design and the methods are scientifically sound. The manuscript is clear and well written. Yet, I have some comments listed thereafter.
Detailed remarks:
Abstract
Line 34. It should be mentioned in the abstract that the CuSO4_10 condition did not induce mortality, what allows the statement that “the toxicity of Cu in the form of CuPT was much higher than that of ionic Cu from CuSO4” (line 40).
Introduction
Line 67-69. As mentioned by authors, very little research has focused on sublethal effects and responses of organisms to CuPT exposure. However, some references provide information on embryonic toxicity and development alterations when fish are exposed to this biofouling, that authors should add to their manuscript (for instance Shin et al., 2022. Ecotoxicology and Environmental Safety, 233,113337; Almond and Trombetta, 2017, Ecotoxicology, 26(7), pp. 855-867; Almond and Trombetta, 2016, Ecotoxicology, 25(2), pp. 389-398).
Material and method
Lines 99-105. All this information on chemical preparation is not relevant without precision on concentrations of the different stock solutions. Were these solutions acidified?
Line 105-106. One would wonder if water quality parameters were measured before or after water renewal, what is explained p7, line 140. It should be mentioned earlier.
Line 112. How long did thermal and environmental acclimation last?
Line 139. “were used for all three analyses” instead of “were used or all”
Line 207. The mean housekeeping genes: was it a geometric or arithmetic mean?
Line 208. N=12 while authors indicate 15 fish per condition (line 138). This should be cleared up.
Results
Figure 1 caption is not clear for Cu analyses (A). Does it correspond to the mean of values just after contamination and 12h after (only one value for both)?
Line 258. These are not the conditions that didn’t accumulate, but the fish exposed to these conditions. Text is quite confused in this section because of not-shown results in gills of the first experiment that are presented within those of the second experiment.
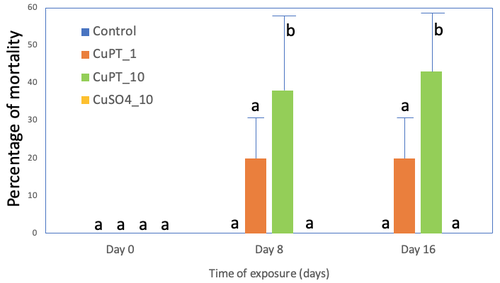
Line 278. Authors write that there was 11% of mortality after 16 days of exposure to cuPT_1 but figure 3 seems to show around 20%.
Lines 284-288. It is quite surprising to present biometric parameters (length and mass) with highest and lowest values of +/- SD each. Why not only means +/- SD?
Lines 290-301. Figures 4B and 4C clearly have been inverted when compared to the text. It seems that 4B presents SOD instead of GPx and vice versa, as written in the caption.
Table 2 and related text are not always consistent:
Line 308. mt1x, mt2x and AcoAc were overexpressed in gills at day 8 of CuPT_10 exposure.
Line 311. ctr1 was not repressed but overexpressed.
Line 317. cox was not repressed in gills at day 16 when exposed to CuPT_1.
Discussion
Same comments as for introduction could be made concerning information on CuPT toxicity in fish embryos provided by scientific literature.
Line 367. Authors consider that differential gene expressions may explain the difference in Cu accumulation in the liver. They should moderate this statement considering that gene expression modulations are not always coupled to modifications of functionally relevant molecules level or activity. Same remark applies for line 466.
Line 431 to 439. Expression results of genes encoding antioxidant enzymes are presented again, but not really discussed. Authors should deepen this section.
Reviewed by Anne-Sophie Voisin, 02 Jun 2023
In their manuscript, Bourdon and co-authors report on experiments aimed at determining the bioaccumulation potential and toxicity of copper pyrithione (CuPT), an alternative antifouling agent to the banned tributyltin, in juvenile rainbow trout. First, a 24h exposure was done with high Cu concentrations from either CuSO4 or CuPT to assess acute toxicity (mortality) and Cu concentration in tissues. Then a second experiment was carried out with lower concentrations to assess chronic exposure to Cu from CuSO4 or CuPT during 8 and 16 hours. In this second experiment, mortality, growth, accumulation of copper, activities of antioxidant enzymes and gene expression were measured. CuPT caused significant mortality both in the acute and chronic exposure, and had a higher toxicity and bioaccumulation potential compared to CuSO4. Although no differences between treatments were observed in enzymatic activities, there was a response at the gene expression level, which was time-dependent.
General comments:
The manuscript is generally very clear and well written. In some instances (see specific comments below), some sentences should be reformulated in a more scientific way. The experimental design and data analysis appear appropriate, and the experiments soundly conducted. The results are well presented and figures and tables are clear and informative. The discussion and conclusions are well supported by the data and answer the original question of the study.
Novelty and relevance: the methods and experimental design do not bring much novelty, however, the toxicity of copper pyrithione is not well known yet and is important to assess. The measure of gene expression at two different timepoints is interesting and shows a time-dependent response.
Specific (minor) comments:
· How were the concentrations chosen?
· 49: suggestion: “causes has … consequences”
· 54: More information on tribulyltin – in which way is it toxic (just in a few words)
· 57:Why/how is PT making copper more bioavailable? Can you mention briefly the modes of action of copper and pyrithione in the introduction? Why is addition of pyrithione important? Is CuPT also used in the paint? Is PT the co-biocide ? This might be evident for readers that know about that compound but otherwise some precisions could be useful.
· 99: please provide the Chemical Abstracts Service (CAS) numbers for the chemicals and where you purchased them
· 102: Is the exposure water tap water or distilled water?
· 106: if the compound is supposed to be rapidly degraded by light, is renewing ¾ of the exposure medium enough?
· 106: what are the outcomes of the water quality parameters? Were they stable and in an acceptable range?
· Are there known degradation products, can they be measured, and do you expect toxicity from those products?
· 120: environmental parameters: could you be more specific
· 135: briefly mention why you chose to analyse the liver and gills for gene expression
· 142: do you provide the results or an estimation for photodegradation?
· 160: why atrazine?
· 183: 500 ml or rather microliters?
· 200: containing?
· 207: against the mean of the housekeeping genes actb and rpl7?
· 207: how were the housekeeping genes selected? Did you verify that there was no effect of the treatments on their expression?
· 207: you get only one Ct value per gene and sample. But you do not get changes in Ct values, right? Or what do you mean by changes ? or do you mean differences in Ct values?
· 208: n=12 but you had 15 individuals (line 181)?
· 211: include this information in the next paragraph about statistical analysis.
· Did you test the efficiency of the primers?
· 216: did you try transformation of the data? Did you apply a correction for multiple testing?
· 221: Afterwards you mention 50 percent of the values between … and … . Could you write this information for the controls (instead of “generally”)?
· 229: with a yield of 50 % with 50 % of the yield?
· 235: Why are Cu concentrations after contamination and 12h after pooled in the same boxplot and not shown separately as for CuPT?
· 242: mention which compounds. Or do you mean concentration? I only see CuPT in Fig1B
· 288: did you calculate the condition factor (although I do not expect to see an effect either if there was no effect on size and weight) ?
· 298: it seems B and C are switched in the legend compared to the graphs? I see a significant reduction in the activity at day 8 in the fig 4B (GPx) but in the text you mention SOD.
· 312: “tended to be more like”, but on a fewer genes… please reformulate, not very clear/scientific.
· 316: ctr1 exposed to CuPT_10?
· 330: is it really a threshold if you test only two or three concentrations? – you cannot calculate a LC50 or EC50 as mentionned at line 393
· 377: like that filtered by the gills: reformulate
· 411: reformulate sentence with marine medaka
· 418: trap, eliminate, increase too much: maybe reformulate in a more scientific language
· 459: molecularly, molecular reponse: repetition. Maybe: …that Cu has induced a molecular response
· 468-469, 477: the gills were able to adapt and defend themselves, … “quite” different: reformulate in more scientific language










